Abstract
Background:
Primary central nervous system lymphoma (PCNSL) is potentially curable when high-dose methotrexate-based induction is followed with intensive consolidation, which can consist of non-myeloablative chemotherapy (NMA-C) or myeloablative chemotherapy (MAC) followed by autologous hematopoietic cell transplantation (autoHCT). Although the efficacy of autoHCT has been demonstrated, very limited data is available regarding the optimal timing of autoHCT, i.e., to transplant in first complete remission (CR1) or second or later CR (CR2+). Moreover, the choice of consolidation differs between centers. Recently, the CALGB 51101 trial showed that MAC leads to improved progression-free survival (PFS) compared to NMA-C, but limiting this finding are that more patients who were randomized to receive NMA-C went off treatment prior to consolidation, either as a result of progression or death. To clarify the optimal timing of autoHCT in PCNSL and to support the findings of the CALGB 51101 trial, we conducted a multi-center retrospective analysis.
Methods:
We retrospectively analyzed patients ≥18 years treated at the University of California campuses in Davis, Fresno, Irvine, and Los Angeles who were diagnosed between January 2005-July 2021. Patients were divided into three cohorts: cohort A included those who underwent autoHCT in CR1, cohort B included those who underwent autoHCT in CR2+, and cohort C included those who underwent NMA-C in CR1 (irrespective of whether or not they underwent autoHCT in CR2+). In the first set of analyses, outcomes were compared between cohort A and B, and in the second set, outcomes were compared between cohort A and C. CR was defined by the lack of enhancement on magnetic resonance imaging. Patients in cohort A and B who were not in CR at the time of autoHCT, and those in cohort C not in CR at the time of NMA-C, were excluded. The primary endpoint was 2-year overall survival (OS), and secondary endpoints were 2-year progression-free survival (PFS), non-relapse mortality (NRM) and cumulative incidence of relapse (CIR). Time-to-event analyses were conducted using the Kaplan-Meier method and were compared by a log-rank test. NRM and CIR were calculated through a competing risk model, with NRM serving as a competing risk for CIR and vice versa. Univariate analyses were performed via Cox proportional hazards model. Patients alive at the end of the study were censored at their last follow-up date.
Results:
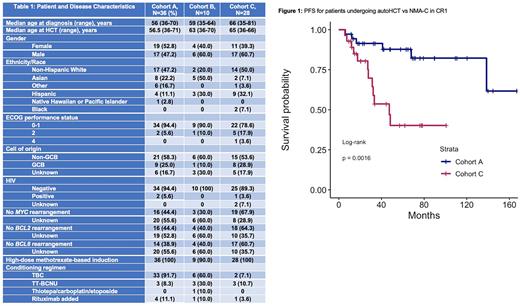
69 unique patients were included, and their baseline characteristics are described in Table 1. 36 patients were included in cohort A, 10 in cohort B, and 28 in cohort C, 5 of whom underwent autoHCT in CR2+. The median follow-up times of cohorts A, B, and C were 67, 73, and 52 months, respectively.
The 2-year OS from the time of diagnosis in cohort A was 94.3% (95% CI, 86.9-100) vs 90.0% (95% CI, 73.2-100) in cohort B (P=0.42), and 84.0% (95% CI, 70.7-99.8) in cohort C (P=0.072). The 2-year PFS from the time of transplant in cohorts A vs B were 91.2% (95% CI, 82.2-100) vs 67.5 (95% CI, 43-100) (P=0.008). The 2-year PFS from the time of diagnosis in cohorts A vs C were 91.4% (95% CI, 82.6-100) vs. 80.5% (95% CI, 66.4-97.6) (P=0.0016) (Figure 1). The 2-year NRM and CIR for cohorts A vs B were 5.8% vs 21.2% (P=0.179) and 2.9% vs 11.3% (P=0.037), respectively. The 2-year NRM and CIR for cohorts A vs C were 5.8% vs 3.6% (P=0.74) and 2.9% vs 16.0% (P<0.001) respectively. Univariate analyses including all 69 unique patients were performed for OS and PFS, and there were no significant associations based on age <60 vs ≥60 years, gender, ECOG performance status ≤1 vs ≥2, center of origin, and thiotepa/busulfan/cyclophosphamide (TBC) vs thiotepa/carmustine (TT-BCNU) conditioning.
Conclusion:
Although limited by the sample size and retrospective nature, whether autoHCT for PCNSL was performed in CR1 or CR2+ did not impact OS in our analysis, but CIR was higher and PFS was lower when autoHCT was performed in CR2+. Supporting the Alliance trial, autoHCT in CR1 produced longer PFS than NMA-C and patients deemed appropriate candidates for MAC consolidation in CR1 should be considered for autoHCT in this setting.
Disclosures
Abdulhaq:Morphosys: Membership on an entity's Board of Directors or advisory committees, Research Funding; Oncopeptides: Speakers Bureau; Alexion: Speakers Bureau; Amgen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BMS: Membership on an entity's Board of Directors or advisory committees; Novartis: Membership on an entity's Board of Directors or advisory committees; Jansen: Membership on an entity's Board of Directors or advisory committees; Genentech: Membership on an entity's Board of Directors or advisory committees. Brem:KiTE Pharma: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Membership on an entity's Board of Directors or advisory committees; Bayer: Membership on an entity's Board of Directors or advisory committees; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; TG Therapeutics: Consultancy; SeaGen: Speakers Bureau; Morphosys/Incyte: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pharmacyclics/Janssen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; BeiGene: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. Heyman:Oncternal Therapeutics, INC: Research Funding; Beigene: Honoraria; Epizyme, INC: Honoraria; Aztrazeneca: Research Funding. Tuscano:ADC therapeutics: Research Funding; Achrotech: Research Funding; BMS: Research Funding; Celgene: Research Funding; Genentech: Research Funding; Takeda: Research Funding; Pharmacyclics: Research Funding.
OffLabel Disclosure:
Carmustine and busulfan are FDA approved for relapsed/refractory non-Hodgkin lymphoma and not necessarily for consolidation.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal